A short reflection on the last day of the Church Year Sunday, 23 November 2008 Reuters report that the Vatican propaganda paper, Osservatore Romano, has stated that the Vatican (aka the Pope) has forgiven John Lennon his 1966 declaration that “the Beatles now are more famous than Jesus Christ”. I wonder what John Lennon would have said about this resurrection from the high prelate in Rome! I suspect at least he would not have liked the Vatican’s motivation: "The remark by John Lennon, which triggered deep indignation ..., after many years sounds only like a 'boast' by a young working-class Englishman faced with unexpected success”. Or is it a ploy by the Vatican when celebrating the last Sunday of the Church Year, the End of the World? ZenMaster
Sunday 23 November 2008
Friday 21 November 2008
Pure Insulin-producing Cells Produced from ESCs
Cells effective in treating diabetes in lab model Friday, 21 November 2008 Singapore researchers have developed an unlimited number of pure insulin-producing cells from mouse embryonic stem cells (ESCs). These pure insulin-producing cells, which according to electron microscopy studies, have the same sub-cellular structures as the insulin-producing cells naturally found in the pancreas, were highly effective in treating diabetes in the mouse model. The transplants of pure insulin-producing cells reduced the blood glucose levels of diabetic mice with high blood glucose levels. The experiments also showed that the subsequent removal of the transplanted cells from the diabetic mice restored the blood glucose to its original high level. None of the diabetic mice involved in the transplant experiments developed teratoma, which are a type of tumour often associated with ESCs and which could complicate their use in human therapeutic treatment. Furthermore, the pure insulin-producing cells managed to retain their insulin-production and glucose-sensing capacity over time. The Singapore researchers' achievement provides proof of principle that this strategy could be applied to human ESCs to obtain similar pure insulin-producing cells. These research findings were published in two separate papers in the July and August 2008 online versions of the journal Stem Cell Research. Conducting the research were scientists at the Institute of Medical Biology (IMB), which is under Singapore's Agency for Science, Technology and Research (A*STAR), and the Yong Loo Lin School of Medicine (YLLSoM) at the National University of Singapore (NUS). The team of researchers was co-led by Dr. Lim Sai Kiang, an IMB principal investigator and a research associate professor at the YLLSoM Department of Surgery, and Dr. Li Guodong, a research associate professor at National University Medical Institutes, YLLSoM, NUS. Commenting about these findings, Dr. Gordon Weir, Director of the Clinical Islet Transplantation Program at Harvard Medical School, who also holds appointments at the Harvard Stem Cell Institute and Joslin Diabetes Centre, said: "The amount of careful work done by this group of researchers is impressive. We need something to put into diabetic patients to treat their condition, and these findings tell us interesting things about the development of beta cells." The strategic approach by the group offers avenues for further research in the treatment for diabetes. "Our ability to isolate and then multiply insulin-producing cells from differentiating ESCs provides an unlimited supply of pure insulin-producing cells to study in unprecedented detail many aspects of these cells," said Dr. Lim. "Besides providing a tool to facilitate basic research in test tubes and animals, these insulin-producing cells may be also used to replace the isolated native pancreatic cells that are hard to obtain in a large amount, for pharmacological tests," added Dr Li. The research was supported primarily by grants from A*STAR's Biomedical Research Council, Juvenile Diabetes Research Foundation International, and National Medical Research Council of Singapore. References: Generating mESC-derived insulin-producing cell lines through an intermediate lineage-restricted progenitor line Li GD, Luo R, Zhang J, Yeo KS, Lian Q, Xie F, Tan EKY, Caille D, Kon OL, Salto-Tellez M, Meda P, and Lim SK Stem Cell Research 2008, in press and available online 8 August 2008. Derivation of functional insulin-producing cell lines from primary mouse embryo culture Li GD, Luo R, Zhang J, Yeo KS, Xie F, Tan EKY , Caille D, Que J, Kon OL, Salto-Tellez M, Meda P, and Lim SK Stem Cell Research 2008, in press and available online 31 July 2008. ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Friday, November 21, 2008
Labels: diabetes, differentiation, embryonic, mouse, research, stem cells 0 comments
Thursday 20 November 2008
Tissue Engineering for Transplanting from Own Stem Cells II
Use of several types of adult stem cells grow new trachea Thursday, 20 November 2008 The first tissue-engineered trachea (windpipe), utilising the patient's own stem cells, has been successfully transplanted into a young woman with a failing airway. The bioengineered trachea immediately provided the patient with a normally functioning airway, thereby saving her life. These remarkable results provide crucial new evidence that adult stem cells, combined with biologically compatible materials, can offer genuine solutions to other serious illnesses. In particular, the successful outcome shows it is possible to produce a tissue-engineered airway with mechanical properties that permit normal breathing and which is free from the risks of rejection seen with conventional transplanted organs. The patient has not developed antibodies to her graft, despite not taking any immunosuppressive drugs. Lung function tests performed two months after the operation were all at the better end of the normal range for a young woman. The pan-European team from the universities of Barcelona, Bristol, Padua and Milan report on this pioneering work in an article published early online and in an upcoming edition of The Lancet. The loss of a normal airway is devastating, but previous attempts to replace large airways have met serious problems. The 30-year-old mother of two, suffering from collapsed airways following a severe case of TB, was hospitalised in March 2008 with acute shortness of breath rendering her unable to carry out simple domestic duties or care for her children. The only conventional option remaining was a major operation to remove her left lung, which carries a risk of complications and a high mortality rate. Based on successful laboratory work previously performed by the team, and given the urgency of the situation, it was proposed that the lower trachea and the tube to the patient's left lung (bronchus) should be replaced with a bioengineered airway based on the scaffold of a human trachea. A seven-centimetre tracheal segment was donated by a 51-year-old transplant donor who had died of cerebral haemorrhage. Spain has a policy of assumed consent for organ donation. Using a new technique developed in Padua University, the trachea was de-cellularised over a six-week period so that no donor cells remained. Stem cells were obtained from the recipient's own bone marrow, grown into a large population in Professor Martin Birchall's lab at the University of Bristol, and matured into cartilage cells (chondrocytes) using an adapted method originally devised for treating osteoarthritis by Professor Anthony Hollander at the University of Bristol. The donor trachea was then seeded with chondrocytes on the outside, using a novel bioreactor which incubates cells, developed at the Politecnico di Milano, Italy, allowing them to migrate into the tissue under conditions ideal for each individual cell type. In order to replicate the lining of the trachea, epithelial cells were seeded onto the inside of the trachea using the same bioreactor. Four days after seeding, the graft was used to replace the patient's left main bronchus. Professor Paolo Macchiarini of the University of Barcelona performed the operation in June 2008 at the Hospital Clínic, Barcelona. Professor Macchiarini, lead author on the paper, said: "We are terribly excited by these results. Just four days after transplantation the graft was almost indistinguishable from adjacent normal bronchi. After one month, a biopsy elicited local bleeding, indicating that the blood vessels had already grown back successfully". Martin Birchall, Professor of Surgery at the University of Bristol, added: "Surgeons can now start to see and understand the very real potential for adult stem cells and tissue engineering to radically improve their ability to treat patients with serious diseases. We believe this success has proved that we are on the verge of a new age in surgical care". Anthony Hollander, Arthritis Research Campaign Professor of Rheumatology and Tissue Engineering at the University of Bristol, concurred: "This successful treatment manifestly demonstrates the potential of adult stem cells to save lives". The patient, Claudia Castillo, a young woman from Colombia but now living in Spain, had no complications from the operation and was discharged from hospital on the tenth post-operative day. She has remained well since and has a normal quality of life. She is able to care for her children, walk up two flights of stairs and occasionally go out dancing in the evenings. She said: "Above all I would like to thank Dr. Macchiarini and his medical team who did the research, for the time and dedication they devoted to my case to make sure that everything turned out alright." Reference: Clinical transplantation of a tissue-engineered airway Paolo Macchiarini, Philipp Jungebluth, Tetsuhiko Go, M Adelaide Asnaghi, Louisa E Rees, Tristan A Cogan, Amanda Dodson, Jaume Martorell, Silvia Bellini, Pier Paolo Parnigotto, Sally C Dickinson, Anthony P Hollander, Sara Mantero, Maria Teresa Conconi, Martin A Birchall The Lancet, Early Online Publication, 19 November 2008, doi:10.1016/S0140-6736(08)61598-6 See also: Tissue Engineering for Transplanting from Own Stem Cells I CellNEWS - Thursday, 20 November 2008 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Thursday, November 20, 2008
Labels: biotech, bone marrow, differentiation, EU, human, medicine, research, stem cells 0 comments
Tissue Engineering for Transplanting from Own Stem Cells I
First tracheal transplant without immunosuppression Thursday, 20 November 2008 Summary:
After 4 years of going from consultation to consultation, Claudia Castillo finally found a solution to her respiratory problems. The young Colombian woman suffered from a cough that took a long time to be diagnosed as tuberculosis. She arrived at
Hospital Clínic of Barcelona with complications and there, she met Professor Paolo Macchiarini, Head of the Thoracic Surgery Department, who led the international team that made possible the first trachea transplant and the first tissue transplant without immunosuppression. She underwent an operation on the upper part of the trachea but nothing could be done to repair the blockage in the left lung. The infection had led to a severe collapse just before the branch of the trachea and this obstruction prevented air from reaching the lung. The only treatment option at the time involved removing the affected lung. As the young mother of two children, removing the lung would have considerably reduced quality of life for Claudia Castillo. In March 2008, her situation worsened to the point where she was unable to carry out domestic chores or look after her children, so intervention became urgent. In June, after obtaining authorization from the ethics committee of Hospital Clínic of Barcelona and from the Catalan Transplant Organization (OCATT), the first trachea transplant and the first tissue transplant of any kind without immunosuppression took place. The study, published online on Wednesday by the journal The Lancet, with Professor Paolo Macchiarini as the principal author, together with his colleagues Dr. Philip Jungebluth, Dr. Tetsuhiko Go and Dr. Jaume Martorell, presents the details of this transplant – the first treatment alternative for treating the collapsed trachea that the patient was suffering from. The technique consists of depleting the trachea to be transplanted of the donor's cells and repopulating it with cells from the recipient before the operation. Thus, thanks to tissue bioengineering, the donor trachea becomes a hybrid that the recipient's body identifies as its own, thereby making immunosuppression unnecessary. The transplant and most of the processes involved were carried out at Hospital Clínic of Barcelona, but this would have been impossible without the collaboration of the University of Bristol (UK), the University of Padua (Italy) and the University of Milan (Italy). Professor Paolo Macchiarini led the prior basic research. The process of preparing the trachea requires many cycles of washing to eliminate all the donor cells – many more than those suggested by the basic research. The tissue was a 7-cm segment of trachea from a 51-year-old donor who had died from brain haemorrhage. The team of Dr. Maria T. Conconi at the University of Padua (Italy) confirmed that, after 25 washing cycles, the trachea treated at Hospital Clínic was free from donor antigens – the molecules that would cause the tissue to be rejected by the recipient. Meanwhile, at the University of Bristol, the teams of Professor Martin Birchall and Professor Anthony Hollander cultivated the recipient's cells that would later be introduced into the trachea. These cells were epithelial cells taken from the trachea and cartilage cells (chondrocytes), differentiated from stem cells taken from the patient's bone marrow. This technique was initially designed to treat cases of osteoarthritis. Back at Hospital Clínic, the team of Professor Paolo Macchiarini introduced these cells into the trachea using a bioreactor designed by the team of Dr. Sandra Mantero at the University of Milan. The epithelial cells were inserted into the inner surface of the trachea and the chondrocytes covered the outer surface. The donor tissue thus became a hybrid very similar to new tissue from the patient herself. The operation was performed 4 days later at Hospital Clínic, where the thoracic surgery team extracted the damaged section of trachea and replaced it with the new trachea. This pioneering operation was not without question marks but if anything had gone wrong, it would have been changed to a lung-resection operation – the classical treatment choice. Thanks to the skill of the surgeons and the huge international effort, the operation was a success. Five months later, the lung that had been so long out of use was providing normal respiration. This innovation in biomedicine and surgery may become an alternative for diseases of the upper airways, such as congenital deformities or primary tumours, which cannot currently be treated using conventional surgical techniques. The clinical application of stem cell cultures and the prevention of the problems deriving from immunosuppression are a milestone in the history of transplantation. There are already some cases being studied that may benefit from the new technique and research continues into improving the process. If all goes well, Claudia Castillo will be just the first patient to benefit from a new advance led by researchers from Hospital Clínic of Barcelona. Reference: Clinical transplantation of a tissue-engineered airway Paolo Macchiarini, Philipp Jungebluth, Tetsuhiko Go, M Adelaide Asnaghi, Louisa E Rees, Tristan A Cogan, Amanda Dodson, Jaume Martorell, Silvia Bellini, Pier Paolo Parnigotto, Sally C Dickinson, Anthony P Hollander, Sara Mantero, Maria Teresa Conconi, Martin A Birchall The Lancet, Early Online Publication, 19 November 2008, doi:10.1016/S0140-6736(08)61598-6See also: Tissue Engineering for Transplanting from Own Stem Cells II CellNEWS - Thursday, 20 November 2008 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Thursday, November 20, 2008
Labels: biotech, bone marrow, differentiation, EU, human, medicine, research, stem cells 0 comments
Extinct Woolly-mammoth Genome Sequenced
Thursday, 20 November 2008

Scientists at Penn State are leaders of a team that is the first to report the genome-wide sequence of an extinct animal, according to Webb Miller, professor of biology and of computer science and engineering and one of the project's two leaders. The scientists sequenced the genome of the woolly mammoth, an extinct species of elephant that was adapted to living in the cold environment of the northern hemisphere. They sequenced four billion DNA bases using next-generation DNA-sequencing instruments and a novel approach that reads ancient DNA highly efficiently.
"Previous studies on extinct organisms have generated only small amounts of data," said Stephan C. Schuster, Penn State professor of biochemistry and molecular biology and the project's other leader.
"Our dataset is 100 times more extensive than any other published dataset for an extinct species, demonstrating that ancient DNA studies can be brought up to the same level as modern genome projects."

The researchers suspect that the full woolly-mammoth genome is over four-billion DNA bases, which they believe is the size of the modern-day African elephant's genome. Although their dataset consists of more than four-billion DNA bases, only 3.3 billion of them — a little over the size of the human genome — currently can be assigned to the mammoth genome. Some of the remaining DNA bases may belong to the mammoth, but others could belong to other organisms, like bacteria and fungi, from the surrounding environment that had contaminated the sample. The team used a draft version of the African elephant's genome, which currently is being generated by scientists at the Broad Institute of MIT and Harvard, to distinguish those sequences that truly belong to the mammoth from possible contaminants.
"Only after the genome of the African elephant has been completed will we be able to make a final assessment about how much of the full woolly-mammoth genome we have sequenced," said Miller.
The team plans to finish sequencing the woolly mammoth's genome when the project receives additional funding.
 Ball of permafrost-preserved mammoth hair containing thick outer-coat and thin under-coat hairs. Credit: Stephan Schuster lab, Penn State University.
Ball of permafrost-preserved mammoth hair containing thick outer-coat and thin under-coat hairs. Credit: Stephan Schuster lab, Penn State University.
The team sequenced the mammoth's nuclear genome using DNA extracted from the hairs of a mammoth mummy that had been buried in the Siberian permafrost for 20,000 years and a second mammoth mummy that is at least 60,000-years-old. By using hair, the scientists avoided problems that have bedevilled the sequencing of ancient DNA from bones because DNA from bacteria and fungi, which always are associated with ancient DNA, can more easily be removed from hair than from bones. Another advantage of using hair is that less damage occurs to ancient DNA in hair because the hair shaft encases the remnant DNA like a biological plastic, thus protecting it from degradation and exposure to the elements.

Members of the team previously ruled out humans as a cause of extinction for at least one of the Siberian sub-populations — the group appears to have gone extinct at least 45,000 years ago at a time when there were no humans living in Siberia. However, much debate still remains regarding the causes of extinction for the other group and for those populations that lived in other places, such as North America. Currently, the team is searching the mammoth's genome for clues about its extinction.
Posted by ZenMaster at Thursday, November 20, 2008
Labels: DNA, evolution, genome, mammoth, mitochondria, research, sequence, US 0 comments
Tuesday 18 November 2008
Kangaroo Genome Mapped
Kangaroo Genome Mapped Tuesday, 18 November 2008 Australian researchers will today launch the world first detailed map of the kangaroo genome, completing the first phase of the kangaroo genomics project. Researchers at the ARC Centre of Excellence for Kangaroo Genomics (KanGO), including University of Melbourne, ANU, WEHI, University of Sydney, University of NSW and the Australian Genome Research Foundation (AGRF) have built a framework to assemble the genome of a model kangaroo, the tammar wallaby. "A good map is crucial for finding our way around a new genome," said KanGO Director Prof. Jenny Graves, who divides her time between ANU and University of Melbourne. "It enables us to explore how the genome of mammals - including humans - is organized, how it functions, and how it evolved." "Now the world can use information on kangaroo genes and sequences to explore how mammals develop and function," she said. DNA sequence obtained by the Australian Genome Research Facility (AGRF) with funding from the Victorian government will be arranged using the genome map. Researchers say the international race to sequence the genomes of significant species is driven by the power of genome comparisons – particularly of species that are distantly related – to reveal secrets of the genome in humans, as well as other mammals. "Australia's weird and wonderful animals are making crucial contributions," Professor Graves said. "The kangaroo has helped to consolidate Australia's reputation in this important genomics era," she said. Graves says genomic information is extremely powerful. She says KanGO researchers used the kangaroo genome map to solve fundamental genetic puzzles, for instance discovering the gene that controls the sex of a baby, and overturning theories of the origin of our blood proteins. The map and sequence will open up new areas of research into how genes are turned on and off during development of all mammals. "Kangaroos are a marvellous model for studying human development and reproduction because they are born very early and complete much of their development in the pouch - rather than the womb," said Laureate Professor Marilyn Renfree of the University of Melbourne's Zoology Department, who takes over as KanGO Director today. "This makes them a powerful tool for studying the genes and hormones involved in mammalian reproduction and development." Professor Graves says that access to the next generation sequencing technologies will mean that the wealth of genetic information in Australia's native flora and fauna can now be tapped into. "This will provide a depth of understanding never thought possible until recently and lead to new and exciting applications in the field of biotechnology." ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Tuesday, November 18, 2008
Labels: chromosomes, DNA, evolution, research, sequence 0 comments
‘Orphan’ Genes Importance in Evolution
‘Orphan’ Genes Importance in Evolution
Tuesday, 18 November 2008
Closely related animal species share most of their genes and look almost identical. However, minor morphological differences allow us to tell them apart. What is the genetic basis for these differences?
Often, the explanation is provided by minor changes in spatial and temporal activity of transcription factors - "regulator" genes that are conserved throughout the animal kingdom. However, every group of animals also possesses a small proportion of genes, which are, in contrary, extremely variable among closely related species or even unique. For example, a gene may be present in one species or animal group, but not in any other. Such genes are referred to as "novel," "orphan" or "taxonomically restricted". Their function and origin are often obscure. What are these genes needed for?
A new paper, published in this week's issue of the online open access journal, PLoS Biology, explores this question in the freshwater polyp Hydra, which belongs to the same branch of the evolutionary tree as jelly fish. These animals are small (several mm long), predatory creatures, with a tube like body-form that ends in a mouth surrounded by mobile tentacles. They are of particular interest to scientists for their regenerative properties, and because they appear to be biologically immortal; not undergoing the aging process that affects all other known animals.
In this paper, a team of scientists from the Christian-Albrechts-Universität zu Kiel in Germany, used transgenic polyps to uncover the role of "orphan" genes in these morphologically-simple animals. The work, led by Thomas Bosch reports that a family of "novel" genes is responsible for morphological differences between two closely related species of fresh water polyps called Hydra. Their most remarkable finding is that a secreted protein, encoded by "novel" gene Hym301, controls the pattern in which the tentacles in Hydra develop.
"We knew that these genes were important, but it was in no sense simple to demonstrate that," says Konstantin Khalturin, first author of the PLoS Biology paper.
A freshwater polyp Hydra and its tentacles during bud formation. In one species, Hydra oligactis, emergence of its tentacles during bud formation is not synchronised; in Hydra vulgaris all five tentacles develop simultaneously and symmetrically; in Hydra vulgaris polyps genetically altered to produce large amounts of protein from the “orphan gene” Hym301, tentacles are formed in an irregular and asymmetric pattern.
The data indicate that "novel" genes are involved in generation of novel morphological features that characterise different species, thus pointing the way to a new, more complete understanding of how evolution works at the level of a particular group of animals. Emergence of "novel" genes may reflect evolutionary processes, which allow animals to adapt in the best way to changing environmental conditions and new habitats.
Reference:
A novel gene family controls species-specific morphological traits in Hydra
Konstantin Khalturin

For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Tuesday, November 18, 2008
Labels: differentiation, evolution, genome, research 0 comments
US Scientists Self-censor During Bush Administration
US Scientists Self-censor During Bush Administration Tuesday, 18 November 2008 A survey of scientists whose studies became the focus of a public debate about NIH grant funding has found that many of them engaged in self-censorship as a result of the controversy. The study, published in the open access journal PLoS Medicine, found that following the criticism of their research, scientists removed politically sensitive language from grant applications and stopped studying certain topics. These self-censorship tactics were employed despite the fact that all of the criticised studies — most of which investigated sexual behaviour, drug-use, and other HIV-related questions — were defended in an NIH internal review and retained their funding. Joanna Kempner of Rutgers University in New Jersey surveyed the researchers who were the subject of a debate in the US in July 2003, which began when a Congressional Representative proposed an amendment to rescind five NIH grants after publicly criticising the studies as "less worthy of taxpayer funding" than research into "devastating diseases." The amendment failed to pass but the controversy resulted in the internal review of more than 250 grants by the NIH, which concluded that each study was scientifically sound. After conducting in-depth interviews with thirty researchers whose funding was reviewed, Kempner surveyed the principal investigators of all the studies. Only a third of the 82 respondents felt they were less likely to receive funding from the NIH in the future, but a majority reported undertaking strategies designed to disguise the most controversial aspects of their research. Half (51%) said they removed potential "red flag" words from the titles and abstracts of their grant submissions, including the words gay, lesbian, homophobia, anal sex, needle-exchange, and AIDS. Kemper reports that one interviewee said "I do not study sex workers, I study 'women at risk.'" Almost a quarter of the researchers had either reframed their studies to avoid research on marginalized or stigmatized populations or had chosen to drop studies that were thought to be political sensitive, such as those on sexual orientation, abortion, childhood sexual abuse, and condom use. The survey also found that four of the principal investigators had made career changes and left academia as a result of the controversy. Joanna Kempner stresses that the controversy also galvanized sections of the research community with 10% of respondents reporting a strengthened commitment to see their research completed, including those who had reported self-censorship practices. She says that the findings are a powerful example of how the political environment can shape what scientists chose not to study. Reference: The chilling effect: How do researchers react to controversy? Kempner J PLoS Med 5(11): e222. doi:10.1371/journal.pmed.0050222 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Tuesday, November 18, 2008
Labels: ethics, human, politics, research, US, US Congress 0 comments
Monday 17 November 2008
Cellular Damage in Huntington's Disease
New clues emerge from study Monday, 17 November 2008 "Huntington's disease presents an ideal vantage point to study neurodegenerative disease, because we know the misfolded protein that's responsible," says Martin Duennwald, formerly a postdoctoral researcher in the lab of Whitehead Institute for Biomedical Research member Susan Lindquist. "But we don't understand how this protein causes cellular damage and death for the neurons that are affected." In a study published in Genes & Development online on November 17, however, Duennwald and Lindquist report the discovery of a mechanism driven by the misfolded proteins that could be one early trigger for cell death. In the U.S., about 1 in 20,000 people suffers from Huntington's. Better understanding of the cellular toxicity may allow new therapies for this fatal and incurable disorder. "This is a diabolical disease, because the misfolded protein interacts with and probably traps many other proteins in the cell," notes Lindquist, who is also a Howard Hughes Medical Institute investigator and a professor of biology at Massachusetts Institute of Technology. Scientists have long known that a single mutated gene that creates proteins with abnormally long repeats of the amino acid glutamine (“Q”) drives Huntington’s. In certain neurons, these "polyQ-expanded" proteins misfold and clump together, damaging and eventually killing the cells. But the steps that kick off the process of cell damage and death have remained a mystery, remarks Duennwald, now a principal scientist at Boston Biomedical Research Institute in Watertown, Mass. In the study, Duennwald first examined what makes polyQ-expanded proteins toxic in yeast. He then performed similar experiments in two kinds of mammalian cells — rat cells that model neurons and mouse striated cells (from the part of the brain most afflicted in Huntington's). He found that cells generated with polyQ-expanded fragments quickly showed problems with proteins that had been marked for degradation in the endoplasmic reticulum (ER, a cell component that folds and finalizes proteins). Such proteins were not expelled for tagging and degradation in the cytosol, the intracellular fluid, outside the ER. "With no garbage disposal, all of a sudden the ER is flooded with proteins that need to be degraded," he says. This breakdown in protein quality control may lead toward cell damage and death. "We were quite surprised because the ER didn't seem to have any connection with the misfolded proteins in the cytosol," Duennwald adds. "This study tells us to investigate cellular pathways beyond the usual suspects." He went on to uncover the basis for this breakdown: The polyQ-expanded fragments glom onto the key VCP/Npl4/Ufd1 protein complex that aids in the transport and degradation of the proteins that flunk quality control in the ER. When Duennwald genetically modified cells to over-express two crucial proteins in the protein complex, the toxic effect dropped. Additionally, his experiments showed that polyQ-expanded proteins avoid a main method by which cells deal with misfolded proteins. Generally, a class of proteins called "chaperone" or "heat shock" proteins move in and either help the misfolded proteins assume their normal shape or help to get rid of them. "Amazingly, polyQ-expanded proteins don't elicit the heat shock response, and that might contribute to their toxicity," Duennwald says. Such findings may help in eventually treating the disease. The research suggests that activating the cell's protein quality control mechanisms may provide novel and effective strategies for combating Huntington's and other illnesses driven by polyQ-expanded proteins. Reference: Impaired ERAD and ER stress are early and specific events in polyglutamine toxicity Martin L. Duennwald and Susan Lindquist Genes & Development, December 2008 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Monday, November 17, 2008
Labels: aging, brain, human, neurons, research, US 0 comments
How Cilia Make Us Asymmetric
FoxJ1 helps cilia beat a path to asymmetry
Monday, 17 November 2008
New work at the Salk Institute for Biological Studies reveals how a genetic switch, known as FoxJ1, helps developing embryos tell their left from their right. While at first glance the right and left sides of our bodies are identical to each other, this symmetry is only skin-deep. Below the surface, some of our internal organs are shifted sideways — heart and stomach to the left, liver and appendix to the right.
Creating this left-right asymmetry is a key step in early embryonic development, and requires hundreds of tiny hair-like structures called nodal cilia to beat in unison. Like microscopic conductors, cilia orchestrate a flow of embryonic fluid from right to left that allows the growing tissues to orient themselves. The current study provides new insight into the crucial role FoxJ1 plays in directing the development of these cilia.
"This one transcription factor regulates a whole suite of genes needed to coordinate the formation of nodal cilia," says Christopher R. Kintner, Ph.D., a professor in the Molecular Neurobiology Laboratory, who led the study. Strikingly, FoxJ1 can induce cilia to form on the surface of cells that do not usually have them, the Salk researchers report in this week's early online edition of Nature Genetics. Their findings may one day lead to a cure for ciliopathies, diseases that result from malfunctioning or damaged cilia.
Cilia — tiny hair-like protrusions found on certain cell types — come in three flavours. Motile cilia crowd the surface of specialized cells and move in harmony to generate liquid flow. They are used to sweep mucus and dirt out of our lungs and in females to propel the egg from the ovary through the Fallopian tube into the uterus. Unlike motile cilia, sensory cilia usually number just one per cell and are used to relay information back to the cell about its surroundings.
A third and less characterized subtype are nodal cilia. Nodal cilia share certain features with both their sensory and motile counterparts; they exist one per cell yet function to generate the fluid movement during embryo development that is crucial to forming the left-right asymmetry.
 In the developing embryo, nodal cilia (shown in green) orchestrate a flow of embryonic fluid from right to left that allows the growing tissues to orientate themselves. Credit: Courtesy of Jennifer Stubbs, Salk Institute for Biological Studies.
In the developing embryo, nodal cilia (shown in green) orchestrate a flow of embryonic fluid from right to left that allows the growing tissues to orientate themselves. Credit: Courtesy of Jennifer Stubbs, Salk Institute for Biological Studies.
"We were interested in the developmental cues that drive the formation of these different cilia subtypes," says Kintner. Clues from previous work in mice persuaded Kintner and his team to take a closer look at cilia in the South African clawed frog Xenopus, a model popular with developmental biologists, and zebrafish.
In mice, FoxJ1 is needed to drive the formation of motile but not sensory cilia. The Salk researchers depleted FoxJ1 in both Xenopus and zebrafish by injecting embryos with morpholinos, synthetic DNA-like structures that bind to nucleic acids and work like dimmer switches to turn down gene expression. When FoxJ1 was turned down, nodal cilia development was disrupted, causing organ displacement and defects in the left-right asymmetry.
The real surprise came when the scientists increased the levels of FoxJ1.
"We started seeing cilia popping up all over the place," says Kintner, "and they were not random subtypes; they looked just like the nodal cilia that form on the cells to generate the embryonic left-right flow."
"These ectopic cilia were really interesting," adds Jennifer Stubbs, first author of the study and a graduate student in the Kintner lab, "and no one had been able to show them in any other system."
These findings call into question current theories as to how FoxJ1 regulates motile cilia. Motile cilia are anchored to the cell surface at sites called basal bodies, and FoxJ1's role in their development was thought to act primarily by regulating this docking process. Since activating FoxJ1 was sufficient to drive the formation of cilia in usually cilia-less cells, however, Kintner and colleagues reasoned that FoxJ1 must play a broader role in promoting cilia development.
They tested this hypothesis using microarray analysis to determine what genes FoxJ1 activated. Indeed, FoxJ1 increased the levels of a host of genes involved in motile cilia development rather than just a small set relating to the basal body.
"This really suggests that at least in Xenopus, FoxJ1 is a master-regulator of ciliogenesis and doesn't just play a role in basal body docking," says Stubbs.
Kintner and colleagues are currently investigating in closer detail the suite of genes activated by FoxJ1 to further understand its mode of action.
“Doing so might help develop novel therapies to treat ciliopathies, whose symptoms range from respiratory defects to infertility. In many diseases such as chronic asthmas and cystic fibrosis, trouble clearing mucus causes defects where the ciliated cells begin to die," says Kintner, "and knowing about the dominant pathways that drive differentiation of ciliated cells types might allow us to do something prevent that situation."
"It may provide a way of repairing ciliated cells that are already there, enabling them to regrow their cilia," says Jennifer Stubbs.
.........
ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Monday, November 17, 2008
Labels: cilia, differentiation, embryo, research, transcription factors, US 0 comments
Tiny Sacs from Cells Carry Information about Tumours
Exosomes deliver factors that promote tumour growth and may serve as blood biomarkers Monday, 17 November 2008 Microvesicles – tiny membrane-covered sacs – released from glioblastoma cells contain molecules that may provide data that can guide treatment of the deadly brain tumour. In their report in the December 2008 Nature Cell Biology, which is receiving early online release, Massachusetts General Hospital (MGH) researchers describe finding tumour-associated RNA and proteins in membrane microvesicles called exosomes in blood samples from glioblastoma patients. Detailed analysis of exosome contents identified factors that could facilitate a tumour's growth through delivery of genetic information or proteins, or signify its vulnerability to particular medications. "Glioblastomas release exosomes in sufficient quantities to pass the blood-brain barrier. We were able to isolate them, analyze the RNA transcripts and show how they might be used as biomarkers to guide targeted therapy and monitor treatment response," says Johan Skog, PhD, the study's lead author, who works in the laboratory of Xandra Breakefield, PhD, at the MGH Neuroscience Center. "Exosomes also may someday be used to deliver therapeutic molecules to the site of a tumour," he added. Many types of cells release exosomes as part of normal cell-to-cell communication, and several types of tumours are known to shed exosomes containing proteins that can alter the cellular environment to favour tumour growth. The current investigation is believed to be the first to carefully analyze the contents of exosomes shed from glioblastoma cells and characterize their contents. The investigators first analyzed tumour cells from three glioblastomas and verified that the cells released exosomes containing RNA and protein molecules. Some messenger RNAs related to activities such as cell proliferation and migration, angiogenesis, and immune response were highly abundant in the exosomes. When glioblastoma exosomes were cultured with normal cells, tumour RNA was delivered into the normal cells and generated its encoded protein, supporting the role of exosome-delivered RNA in manipulating the cellular environment. To study the potential of glioblastoma exosomes as markers of a tumour's genetic makeup, the researchers analyzed tumour tissue and blood serum from 25 glioblastoma patients and were able both to find tumour exosomes and to identify, in some tissue samples, a mutation in the epidermal growth factor receptor (EGFR) gene that characterizes a tumour subtype. In two patients, an EGFR mutation that did not appear in the tumour tissue sample was identified by exosome analysis, reflecting how a surgical biopsy can miss tissue conveying critical information because of the often-chaotic diversity of cells within a tumour. "It is known that the effects of some anticancer drugs depend on a tumour's genetic mutational profile, so our results have broad implications for personalized medicine," explains Skog, who is an instructor in Neurology at Harvard Medical School. "Detecting mutational profiles through a non-invasive blood test could allow us to monitor how a tumour's genetic makeup changes in response to therapy, which may necessitate changes in treatment strategy." Skog, Breakefield and their colleagues are also investigating the role of exosomes in other solid tumours and how they may help monitor additional tumour-associated mutations. The current study was supported by grants from the Wenner-Gren Foundation, Stiftelsen Olle Engkvist Byggmästare, the National Cancer Institute, the Brain Tumour Society and the American Brain Tumour Association. The MGH's provisional patent on the work described in this study has been exclusively licensed to Exosome Diagnostics, Inc.. Subsequent to the completion of this work, Skog was appointed the company's director of Research, while maintaining his position at MGH. ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Monday, November 17, 2008
Labels: Cancer, human, neurons, patent, research, RNA, US 0 comments
Wednesday 12 November 2008
Newborn Neurons in the Adult Brain Can Settle in the Wrong Neighbourhood
Newborn Neurons in the Adult Brain Can Settle in the Wrong Neighbourhood
Wednesday, 12 November 2008
A new study published in this week's PLoS Biology could have significant consequences for neural tissue transplantation to treat brain injuries or neural degeneration. Researchers at the Salk Institute for Biological Studies report that inactivating a specific gene in adult neural stem cells makes nerve cells emerging from those precursors form connections in the wrong part of the adult brain.
In fact, the inappropriate synaptic connections made by cdk5-deficient cells persisted for months after the treatment with cdk5-antagonizing retroviruses.
"One might have predicted that aberrant maturing nerve cells would get kicked out of the circuitry later on," reports Jessberger, who followed the behaviour of newborn granule cells in treated mice long after cdk5 activity was eliminated.
"Even after one year, some of those cells remained in the wrong part of the hippocampus."
"The nice part of this story is that it emerged from a systems genetics approach we used in a previous study," says Gage.
"It continues our effort to apply genetic analysis to find chromosomal regions harbouring genes that may play a critical role in neurogenesis."
Reference:
Cdk5 regulates accurate maturation of newborn granule cells in the adult hippocampus
Sebastian Jessberger, Stefan Aigner, Gregory D. Clemenson Jr., Nicolas Toni, D. Chichung Lie, Özlem Karalay, Rupert Overall, Gerd Kempermann, Fred H. Gage
PLoS Biol (2008) 6(11): e272.  The research team, led by Professor Fred H. Gage, Ph.D., discovered that a protein called cdk5 is necessary for both correct elaboration of highly branched and complex dendrites, a kind of antennae, which are extended by neurons. Cdk5 is also involved in the proper migration of cells bearing those antennae.
Previously described functions of cdk5 are manifold, among them neuronal migration and dendritic path-finding of neurons born during embryonic development.
"The surprising element was that the dendrites of newborn granule cells in the adult hippocampus lacking cdk5 stretched in the wrong direction and actually formed synapses with the wrong cells," explains Gage. Synapses are the specialized contact points where dendrites receive input from the long processes, or axons, of neighbouring neurons. The investigators injected retroviruses into the hippocampus of adult mice to tag and knock out cdk5 activity in newborn granule cell neurons.
These findings offer extremely valuable, unanticipated insight.
"Our data shows that cells that fail to find their 'right spot' might actually become integrated into the brain and possibly interfere with normal information processing," says the study's lead author Sebastian Jessberger, M.D., a former postdoc in the Gage lab and now an assistant professor at the Swiss Federal Institute of Technology in Zurich, Switzerland.
"We found that dendrites of cells lacking cdk5 seemed to integrate into the brain no matter what direction they grew in," he says.
Gage notes that the findings "reflect the need for therapeutic approaches that will assure that cells used in regenerative medicine are strategically placed so that they will make appropriate rather than promiscuous connections."
The research team, led by Professor Fred H. Gage, Ph.D., discovered that a protein called cdk5 is necessary for both correct elaboration of highly branched and complex dendrites, a kind of antennae, which are extended by neurons. Cdk5 is also involved in the proper migration of cells bearing those antennae.
Previously described functions of cdk5 are manifold, among them neuronal migration and dendritic path-finding of neurons born during embryonic development.
"The surprising element was that the dendrites of newborn granule cells in the adult hippocampus lacking cdk5 stretched in the wrong direction and actually formed synapses with the wrong cells," explains Gage. Synapses are the specialized contact points where dendrites receive input from the long processes, or axons, of neighbouring neurons. The investigators injected retroviruses into the hippocampus of adult mice to tag and knock out cdk5 activity in newborn granule cell neurons.
These findings offer extremely valuable, unanticipated insight.
"Our data shows that cells that fail to find their 'right spot' might actually become integrated into the brain and possibly interfere with normal information processing," says the study's lead author Sebastian Jessberger, M.D., a former postdoc in the Gage lab and now an assistant professor at the Swiss Federal Institute of Technology in Zurich, Switzerland.
"We found that dendrites of cells lacking cdk5 seemed to integrate into the brain no matter what direction they grew in," he says.
Gage notes that the findings "reflect the need for therapeutic approaches that will assure that cells used in regenerative medicine are strategically placed so that they will make appropriate rather than promiscuous connections."
 Newborn neurons deficient in cdk5 (green) extend aberrant dendrites that nonetheless synaptically integrate into the pre-existing dentate circuitry containing neurons (red) and glial cells (blue). From: For New Neurons in an Old Brain, cdk5 Shows the Way Robinson R PLoS Biology Vol. 6, No. 11, e291 doi:10.1371/journal.pbio.0060291.
Newborn neurons deficient in cdk5 (green) extend aberrant dendrites that nonetheless synaptically integrate into the pre-existing dentate circuitry containing neurons (red) and glial cells (blue). From: For New Neurons in an Old Brain, cdk5 Shows the Way Robinson R PLoS Biology Vol. 6, No. 11, e291 doi:10.1371/journal.pbio.0060291.
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Wednesday, November 12, 2008
Labels: brain, California, mouse, neurons, stem cells, US 0 comments
Teeth Stem Cells Can Stimulate Growth of Brain Cells in Monkeys
Teeth Stem Cells Can Stimulate Growth of Brain Cells in Monkeys Wednesday, 12 November 2008 Researchers at the Yerkes National Primate Research Center, Emory University, have discovered dental pulp stem cells can stimulate growth and generation of several types of neural cells. Findings from this study, available in the October issue of the journal Stem Cells, suggest dental pulp stem cells show promise for use in cell therapy and regenerative medicine, particularly therapies associated with the central nervous system. Dental stem cells are adult stem cells, one of the two major divisions of stem cell research. Adult stem cells have the ability to regenerate many different types of cells, promising great therapeutic potential, especially for diseases such as Huntington's and Parkinson's. Already, dental pulp stem cells have been used for regeneration of dental and craniofacial cells. Yerkes researcher Anthony Chan, DVM, PhD, and his team of researchers placed dental pulp stem cells from the tooth of a rhesus macaque into the hippocampal areas of mice. The dental pulp stem cells stimulated growth of new neural cells, and many of these formed neurons. "By showing dental pulp stem cells are capable of stimulating growth of neurons, our study demonstrates the specific therapeutic potential of dental pulp stem cells and the broader potential for adult stem cells," says Chan, who also is assistant professor of human genetics in Emory School of Medicine. Because dental pulp stem cells can be isolated from anyone at any age during a visit to the dentist, Chan is interested in the possibility of dental pulp stem cell banking. "Being able to use your own stem cells for therapy would greatly decrease the risk of cell rejection that we now experience in transplant medicine," says Chan. Chan and his research team next plan to determine if dental pulp stem cells from monkeys with Huntington's disease can enhance brain cell development in the same way dental pulp stem cells from healthy monkeys do. Reference: Putative Dental Pulp-Derived Stem/Stromal Cells Promote Proliferation and Differentiation of Endogenous Neural Cells in the Hippocampus of Mice Anderson Hsien-Cheng Huang, Brooke R. Snyder, Pei-Hsun Cheng, Anthony W.S. Chan Stem Cells Vol. 26 No. 10 October 2008, pp. 2654 -2663, doi:10.1634/stemcells.2008-0285 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Wednesday, November 12, 2008
Labels: differentiation, macaque, monkey, neurons, Parkinson, research, stem cells, US 0 comments
Tuesday 11 November 2008
Biological Reaction Essential to Life Takes 2.3B Years Without Enzyme
Biological Reaction Essential to Life Takes 2.3B Years Without Enzyme
Tuesday, 11 November 2008
All biological reactions within human cells depend on enzymes. Their power as catalysts enables biological reactions to occur usually in milliseconds. But how slowly would these reactions proceed spontaneously, in the absence of enzymes – minutes, hours, days? And why even pose the question?
One scientist who studies these issues is Richard Wolfenden, Ph.D., Alumni Distinguished Professor Biochemistry and Biophysics and Chemistry at the University of North Carolina at Chapel Hill. Wolfenden holds posts in both the School of Medicine and in the College of Arts and Sciences and is a member of the National Academy of Sciences.
In 1995, Wolfenden reported that without a particular enzyme, a biological transformation he deemed "absolutely essential" in creating the building blocks of DNA and RNA would take 78 million years.
"Now we've found a reaction that – again, in the absence of an enzyme – is almost 30 times slower than that," Wolfenden said.
"Its half-life – the time it takes for half the substance to be consumed – is 2.3 billion years, about half the age of the Earth. Enzymes can make that reaction happen in milliseconds."
With co-author Charles A. Lewis, Ph.D., a postdoctoral scientist in his lab, Wolfenden published a report of their new findings recently in the online early edition of the Proceedings of the National Academy of Science. The study is also due to appear in the Nov. 11 print edition.
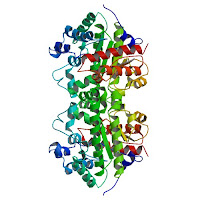 The reaction in question is essential for the biosynthesis of haemoglobin and chlorophyll, Wolfenden noted. But when catalyzed by the enzyme uroporphyrinogen decarboxylase, the rate of chlorophyll and haemoglobin production in cells "is increased by a staggering factor, one that's equivalent to the difference between the diameter of a bacterial cell and the distance from the Earth to the sun."
"This enzyme is essential for both plant and animal life on the planet," Wolfenden said.
"What we're defining here is what evolution had to overcome, that the enzyme is surmounting a tremendous obstacle, a reaction half-life of 2.3 billion years."
Knowing how long reactions would take without enzymes allows biologists to appreciate their evolution as prolific catalysts, Wolfenden said. It also enables scientists to compare enzymes with artificial catalysts produced in the laboratory.
"Without catalysts, there would be no life at all, from microbes to humans," he said.
"It makes you wonder how natural selection operated in such a way as to produce a protein that got off the ground as a primitive catalyst for such an extraordinarily slow reaction."
Experimental methods for observing very slow reactions can also generate important information for rational drug design based on cellular molecular studies.
"Enzymes that do a prodigious job of catalysis are, hands-down, the most sensitive targets for drug development," Wolfenden said.
"The enzymes we study are fascinating because they exceed all other known enzymes in their power as catalysts."
Wolfenden has carried out extensive research on enzyme mechanisms and water affinities of biological compound. His work has also influenced rational drug design, and findings from his laboratory helped spur development of ACE inhibitor drugs, now widely used to treat hypertension and stroke. Research on enzymes as proficient catalysts also led to the design of protease inhibitors that are used to treat HIV infection.
"We've only begun to understand how to speed up reactions with chemical catalysts, and no one has even come within shouting distance of producing, or predicting the magnitude of, their catalytic power," Wolfenden said.
Reference:
Uroporphyrinogen decarboxylation as a benchmark for the catalytic proficiency of enzymes
Charles A. Lewis, Jr. and Richard Wolfenden
PNAS, November 6, 2008, doi: 10.1073/pnas.0809838105
.........
ZenMaster
The reaction in question is essential for the biosynthesis of haemoglobin and chlorophyll, Wolfenden noted. But when catalyzed by the enzyme uroporphyrinogen decarboxylase, the rate of chlorophyll and haemoglobin production in cells "is increased by a staggering factor, one that's equivalent to the difference between the diameter of a bacterial cell and the distance from the Earth to the sun."
"This enzyme is essential for both plant and animal life on the planet," Wolfenden said.
"What we're defining here is what evolution had to overcome, that the enzyme is surmounting a tremendous obstacle, a reaction half-life of 2.3 billion years."
Knowing how long reactions would take without enzymes allows biologists to appreciate their evolution as prolific catalysts, Wolfenden said. It also enables scientists to compare enzymes with artificial catalysts produced in the laboratory.
"Without catalysts, there would be no life at all, from microbes to humans," he said.
"It makes you wonder how natural selection operated in such a way as to produce a protein that got off the ground as a primitive catalyst for such an extraordinarily slow reaction."
Experimental methods for observing very slow reactions can also generate important information for rational drug design based on cellular molecular studies.
"Enzymes that do a prodigious job of catalysis are, hands-down, the most sensitive targets for drug development," Wolfenden said.
"The enzymes we study are fascinating because they exceed all other known enzymes in their power as catalysts."
Wolfenden has carried out extensive research on enzyme mechanisms and water affinities of biological compound. His work has also influenced rational drug design, and findings from his laboratory helped spur development of ACE inhibitor drugs, now widely used to treat hypertension and stroke. Research on enzymes as proficient catalysts also led to the design of protease inhibitors that are used to treat HIV infection.
"We've only begun to understand how to speed up reactions with chemical catalysts, and no one has even come within shouting distance of producing, or predicting the magnitude of, their catalytic power," Wolfenden said.
Reference:
Uroporphyrinogen decarboxylation as a benchmark for the catalytic proficiency of enzymes
Charles A. Lewis, Jr. and Richard Wolfenden
PNAS, November 6, 2008, doi: 10.1073/pnas.0809838105
.........
ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Tuesday, November 11, 2008
Labels: biotech, chemistry, evolution, research, US 0 comments
Umbilical Cord Blood May Help Build New Heart Valves
Umbilical Cord Blood May Help Build New Heart Valves Tuesday, 11 November 2008 Children with heart defects may someday receive perfectly-matched new heart valves built using stem cells from their umbilical cord blood, according to research presented at the American Heart Association's Scientific Sessions 2008. When infants are born with malfunctioning heart valves that cannot be surgically repaired, they rely on replacements from animal tissue, compatible human organ donations or artificial materials. These replacements are lifesaving, but do not grow and change shape as a child develops; so two or more surgeries may be needed to replace outgrown valves. The animal tissue may also stiffen over time as well and be less durable than a normal human valve. With artificial valves, children also must be treated with blood thinners. "In our concept, if prenatal testing shows a heart defect, you could collect blood from the umbilical cord at birth, harvest the stem cells, and fabricate a heart valve that is ready when the baby needs it," said Ralf Sodian, M.D., lead author of the study and a cardiac surgeon at the University Hospital of Munich. The tissue engineering of heart valves is still in its infancy, with various researchers investigating the possibility of using cells from blood, bone marrow or amniotic fluid. In the study, the research team used stem cells (CD133+ cells) derived from umbilical cord blood. The cord blood was frozen to preserve it. After 12 weeks, the cells were seeded onto eight heart valve scaffolds constructed of a biodegradable material and then grown in a laboratory. Afterwards, examination using electron microscopes revealed that the cells had grown into pores of the scaffolding and formed a tissue layer. Biochemical examination indicated that the cells had not only survived and grown, but had produced important elements of the "extracellular matrix," the portion of body tissue that functions outside of cells and is essential to tissue function and structure. Compared with human tissue from pulmonary heart valves, the tissue-engineered valves formed:
- 77.9 percent as much collagen (the main protein in connective tissue);
- 85 percent as much glycosaminoglycan, a carbohydrate important in connective tissue); and
- 67 percent as much elastin (a protein in connective tissue)
Furthermore, using antibodies to detect various proteins, the researchers found the valves contained desmin (a protein in muscle cells), laminin (a protein in all internal organs), alpha-actin (a protein that helps muscle cells contract) and CD31, VWF and VE-cadherin (components of blood vessel linings). "These markers all indicate that human cardiovascular tissue was grown in the lab," Sodian said. Several important questions remain to be solved regarding tissue-engineered functional heart valves, including identifying the optimal scaffold material and learning how to condition the valves in the laboratory so they work properly after being implanted, Sodian said. "Tissue engineering provides the prospect of an ideal heart valve substitute that lasts throughout the patient's lifetime and has the potential to grow with the recipient and to change shape as needed," he said. ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Tuesday, November 11, 2008
Labels: Germany, heart, human, research, stem cells, umbilical cord 0 comments
Mechanism that Regulates the Development of Stem Cells into Neurons
Findings provide insight into potential therapies for neurodegenerative disorders and cancers Tuesday, 11 November 2008 Researchers at the University of Southern California (USC) have identified a novel mechanism in the regulation and differentiation of neural stem cells. Researchers found that the protein receptor Ryk has a key role in the differentiation of neural stem cells, and demonstrated a signalling mechanism that regulates neuronal differentiation as stem cells begin to grow into neurons. The study will be published in the Nov. 11 issue of the journal Developmental Cell. The findings could have important implications for regenerative medicine and cancer therapies, says Wange Lu, Ph.D., assistant professor of biochemistry and molecular biology at the Keck School of Medicine of USC, and the principal investigator on the study. "Neural stem cells can potentially be used for cell-replacement therapy for neurodegenerative diseases such as Alzheimer's and Parkinson's Disease, as well as spinal cord injury," Lu says. "Knowledge gained from this study will potentially help to generate neurons for such therapy. This knowledge can also be used to inhibit the growth of brain cancer stem cells." During brain development, neural stem cells respond to the surrounding environment by either proliferation or differentiation, but the molecular mechanisms underlying the development of neural stem cells and neurons are unclear, Lu notes. Ryk functions as a receptor of Wnt proteins required for cell-fate determination, axon guidance and neurite outgrowth in organisms. Researchers at the Eli and Edythe Broad Center for Regenerative Medicine and Stem Cell Research at USC analyzed sections of the forebrain in animal model embryos to investigate Ryk's function in vivo. They found that during neurogenesis, when neural stem cells start to grow into neurons, Ryk protein is cleaved and translocates to the cell nucleus to regulate neuronal differentiation. This finding is extremely important for understanding the regulation of self-renewal and differentiation of neural stem cells, Lu says. Previous research has shown that Ryk functions as a receptor of Wnt proteins. However, the role of Ryk in neural stem cells and the molecular mechanism of Ryk signalling have not previously been known. "This study will help in our efforts to produce nerve cells from embryonic stem cells, and may lead to the development of new strategies for the repair of the nervous system, using protein or small molecule therapeutic agents," says Martin Pera, Ph.D., director of the Eli and Edythe Broad Center for Regenerative Medicine and Stem Cell Research at USC. Further research is needed to explore how Ryk regulates neuronal gene expression, Lu says. Researchers are now expanding their research to studies of differentiation of human embryonic stem cells into neural stem cells and neurons. These studies are very important for regenerative medicine and drug discovery for therapy of neurodegenerative diseases. Reference: Cleavage of Wnt Receptor Ryk Regulates Neuronal Differentiation during Cortical Neurogenesis Jungmook Lyu, Vicky Yamamoto and Wange Lu. Developmental Cell, Volume 15, Issue 5, 773-780, 11 Nov. 2008, doi:10.1016/j.devcel.2008.10.004 ......... ZenMaster
For more on stem cells and cloning, go to CellNEWS at http://cellnews-blog.blogspot.com/ and http://www.geocities.com/giantfideli/index.html
Posted by ZenMaster at Tuesday, November 11, 2008
Labels: Alzheimer, California, differentiation, neurons, Parkinson, research, stem cells, US, Wnt 0 comments




